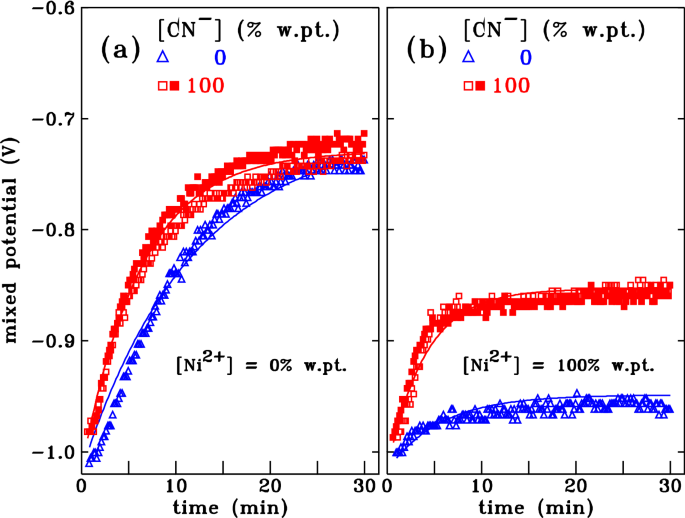
Cyanide removal capacity according to the equilibrium concentration of... | Download Scientific Diagram
Fracture mode: Aluminum wire pull test of flex nickel with cyanide-free... | Download Scientific Diagram
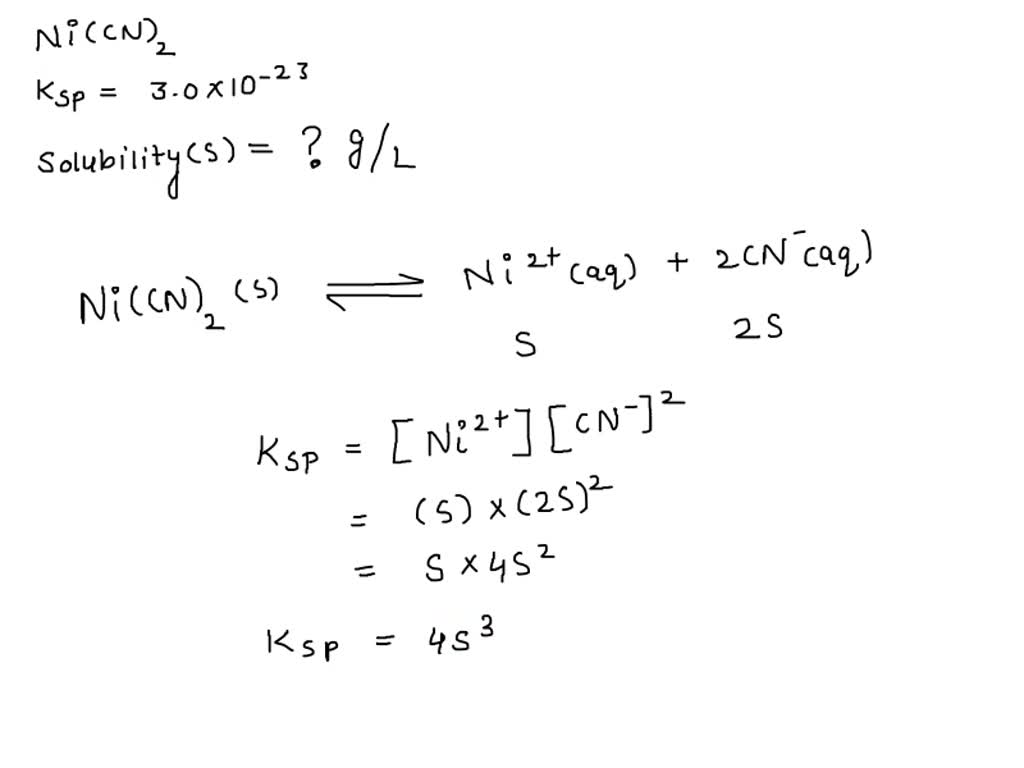
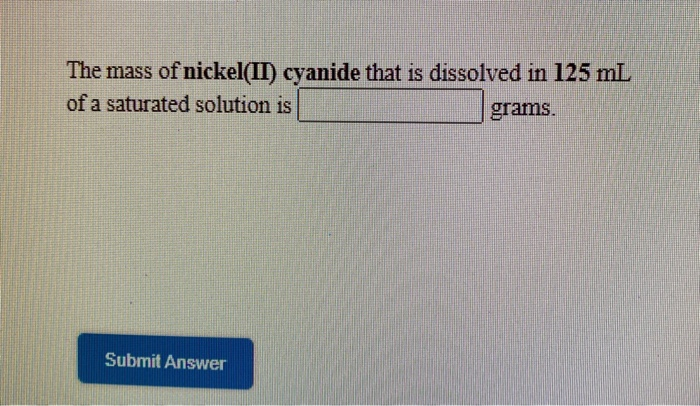
SOLVED: Calculate the solubility of nickel(II) cyanide, Ni(CN)z in units of grams per liter: Ksp(Ni(CN)) = 3.0x10-23. solubility 1.95x104-8 g/L Submit Answer

Flowsheet for the treatment of Nickel/Cobalt residues by dissolution in... | Download Scientific Diagram

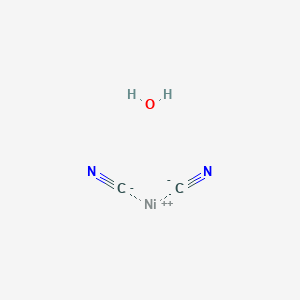
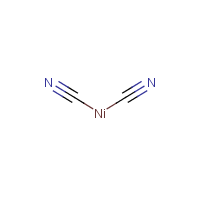
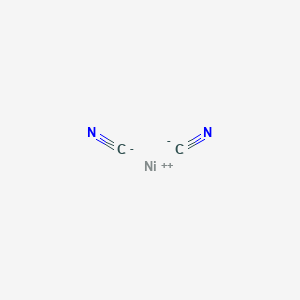
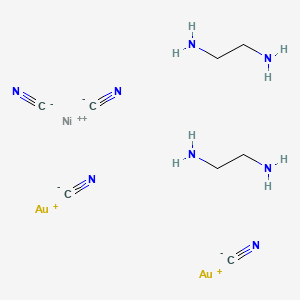
Complexes of nickel(II) cyanide with ditertiary phosphines and their reduction to nickel(I) derivatives - Chemical Communications (London) (RSC Publishing)
Shear strength of flex nickel with cyanide-free versus conventional... | Download Scientific Diagram

SOLVED: Consider the insoluble compound nickel(II) carbonate , NiCO, The nickel ion also forms complex with cyanide ions Write balanced nel ionic equation show why the solubility of NiCO increases in the
Formation of a two-dimensional iron-nickel cyanide-bridged network at... | Download Scientific Diagram