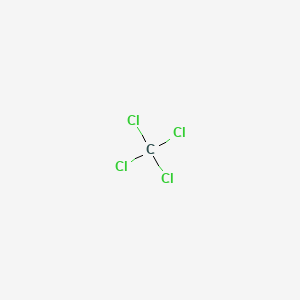
Why is carbon tetrachloride non-polar, while the other tetrachlorides of group four elements are polar? - Quora

Explain why carbon tetrachloride molecules are not polar while carbon trichloride is polar - Digital Teachers Uganda
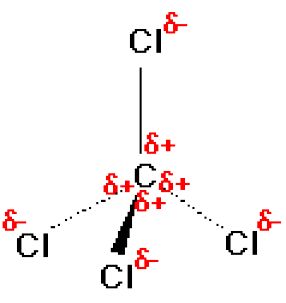
SOLVED: What covalent compound forms when one atom of carbon (C) bonds to four atoms of chlorine (Cl)? carbon tetrachloride carbon tetrachloride carbon chloride carbon chloride tetrachloride carbon tetrachloride carbon monocarbide tetrachloride

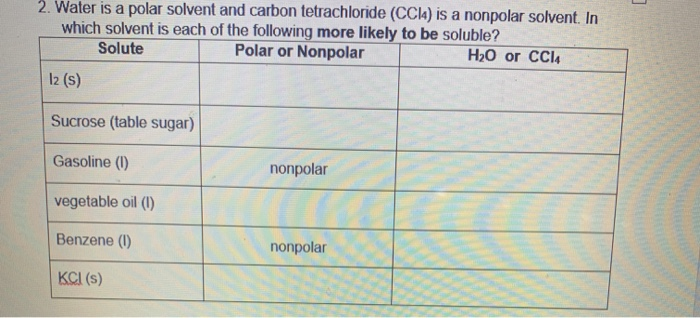
Which solvent, water or carbon tetrachloride, would you choose to dissolve each of the following? a. KrF_2 b. SF_2 c. SO_2 d. CO_2 e. MgF_2 f. CH_2O g. CH_2=CH_2 | Homework.Study.com

Carbon tetrachloride CCl4 lewis dot structure, molecular geometry, polar or nonpolar, Bond angle | Molecular geometry, Molecular shapes, Molecular

Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar

Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar

The carbon tetrachloride molecule (CCl4) is: A) a nonpolar molecule with polar bonds B) a nonpolar molecule with nonpolar bonds C) a polar molecule with polar bonds D) a polar molecule with
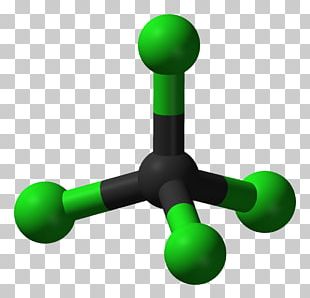
Molecule Molecular Geometry Carbon Dioxide Carbon Tetrachloride Chemical Polarity PNG, Clipart, Carbon, Carbon Dioxide, Carbon Tetrachloride, Chemical Polarity, Chemistry Free PNG Download