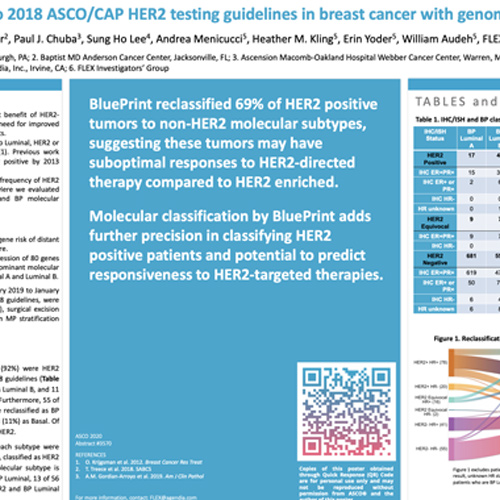
![PDF] HER2 Gene Amplification Testing by Fluorescent In Situ Hybridization (FISH): Comparison of the ASCO-College of American Pathologists Guidelines With FISH Scores Used for Enrollment in Breast Cancer International Research Group Clinical PDF] HER2 Gene Amplification Testing by Fluorescent In Situ Hybridization (FISH): Comparison of the ASCO-College of American Pathologists Guidelines With FISH Scores Used for Enrollment in Breast Cancer International Research Group Clinical](https://d3i71xaburhd42.cloudfront.net/c7289407931d6ff274350f21b4024900edadc66e/2-Figure1-1.png)
PDF] HER2 Gene Amplification Testing by Fluorescent In Situ Hybridization (FISH): Comparison of the ASCO-College of American Pathologists Guidelines With FISH Scores Used for Enrollment in Breast Cancer International Research Group Clinical
Assessment of ERBB2/HER2 Status in HER2-Equivocal Breast Cancers by FISH and 2013/2014 ASCO-CAP Guidelines.,JAMA Oncology - X-MOL

Issues and updates: evaluating estrogen receptor-α, progesterone receptor, and HER2 in breast cancer | Modern Pathology

Estrogen and Progesterone Receptor Testing in Breast Cancer: ASCO/CAP Guideline Update | Journal of Clinical Oncology

Estrogen and Progesterone Receptor Testing in Breast Cancer: ASCO/CAP Guideline Update | Journal of Clinical Oncology

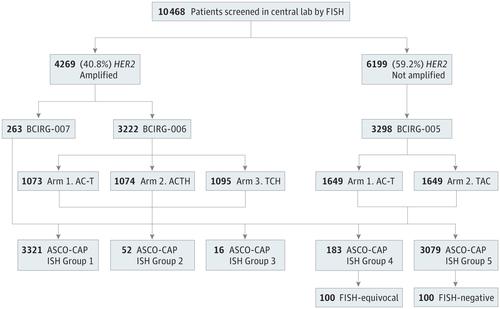
Implementation of the 2018 American Society of Clinical Oncology/College of American Pathologists Guidelines on HER2/neu Assessment by FISH in breast cancers: predicted impact in a single institutional cohort - ScienceDirect
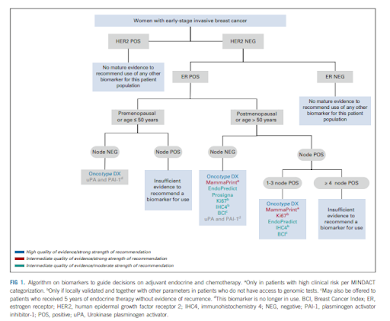
Discoveries in Health Policy: ASCO Updates Guidance for Breast Cancer Prognostics (Adjuvant Chemotherapy)
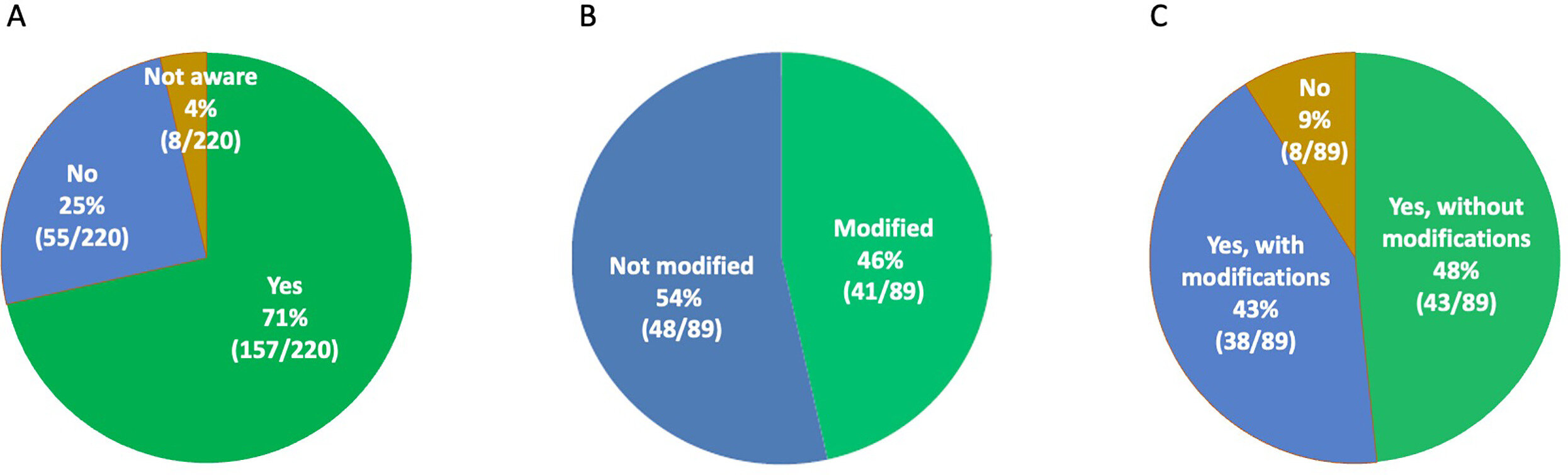
Assessing clinical implementation of past standards for interpretation and reporting of sequence variants in cancer

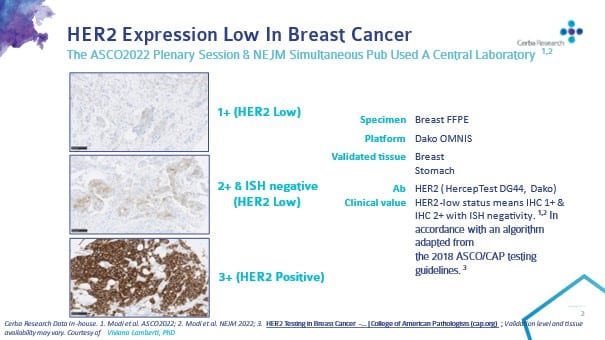
Standardized pathology report for HER2 testing in compliance with 2023 ASCO/ CAP updates and 2023 ESMO consensus statements on HER2-low breast cancer | Virchows Archiv

ASCO/CAP 2018 breast cancer HER2 testing guidelines: summary of pertinent recommendations for practice in Australia - ScienceDirect

Schematic diagram of the ''Algorithm for evaluation of human epidermal... | Download Scientific Diagram

Clinically actionable cancer somatic variants (CACSV): a tumor interpreted dataset for analytical workflows | BMC Medical Genomics | Full Text

Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update | Journal of Clinical Oncology